When last I blogged, the Discovering Insect Species course was in full swing; we were excitedly waiting for the DNA sequence results from our first big field trip, which was to Klamath Marsh National Wildlife Refuge. I’m sorry I didn’t post regular blogs during the remainder of the course, but so many exciting things happened, so quickly, that I never caught my breath enough to write down what was going on. This overview post will be followed by some blog posts about particular aspects of the course, including the discoveries made in the class, some thoughts on teaching gene tree / species delimitation modeling to students, and the class projects, and the Awards Ceremony.
Cutting to the chase, the course was awesome, and tremendously successful. It was the most rewarding experience of my career as a teacher, and I think the students got a huge amount out of it too. We did indeed discover two new species during the course (as promised in the name of the course!), two species I had never seen before, which very much added to the excitement – more on that later.
The course was quite costly, both financially (I spent about $8000 on it) and in terms of my time (and John’s, who served as a TA), but I hope this single experience has a significant impact on the students who participated.
Below is a general overview of the events in the course. There are many details missing here, and I will cover a few of those in later blogs.
Week 1
- Tuesday, 31 March: Introduction. We had exercises with tamarins; students as a group looked at pictures of 26 specimens of tamarins, and had to hypothesize species boundaries. We then did a similar exercise with a subgenus of Bembidion (subgenus Liocosmius, with pictures of the beetles and gene trees).
- Thursday, 2 April: Field trip to the Willamette River in Corvallis where we found four species of Trepanedoris: B. elizabethae, B. fortestriatum, B. siticum, and B. acutifrons.
Week 2
- Tuesday, 7 April: The students learned how to point-mount and label specimens. Students mounted specimens from the previous class’s field trip, and looked at Trepanedoris under the scope, to become familiar with their structures.
- Thursday, 9 April: We used Lindroth’s key to try to identify specimens; this required learning the structures of beetles, and basic terms. The student then sorted pictures of the 43 already-extracted specimens (pictures included habitus, head, genitalia), and in so doing predicted species boundaries. After predictions were made, there was a mini-lecture about the current status of studies of Trepanedoris, including taxonomic history of group. I showed gene trees, and we compared them to the student’s predictions.
- Friday-Sunday: Weekend field trip to Klamath Marsh NWR.
Week 3
- Tuesday, 14 April: Students extracted DNA of specimens from the Corvallis field trip and those from Klamath Marsh NWR. Each student extracted DNA from four specimens, chosen by David. As a whole, the set of specimens studied represent the diversity of forms we collected.
- Thursday, 16 April: Students PCRed 28S and COI for the extracted specimens. After the reactions are on the thermal cyclers, each student point mounts their four voucher specimens, and labels them.
Week 4
- Tuesday, 21 April: Run PCR gels, prepare products for DNA sequencing. While gels are running, we learn how to take pictures of beetles on David’s imaging system.
- Thursday, 23 April: We examined in Google Earth the distribution of previously sequenced Trepanedoris, and the specimens they did genetic work on. Students look at their specimens, and try to identify them. They hand in predictions as to where the sequences of their specimens will fall in the gene trees. Mini-lecture and discussion: what is the Tree of Life?
Week 5
- Monday, 27 April: Plates were sent off to the GATC sequencing facility in Arizona
- Tuesday, 28 April: I showed a slide show of preparing the sequencing plates, and sending them off to be sequenced. Students then took previous sequence data for four genes, and used Mesquite/Zephyr/RAxML to infer gene trees. There was a mini-lecture and exercises about gene trees and species trees and coalescence. There were dice-based exercises, and Mesquite-based coalescent simulations. I’ll write a separate blog about these exercises.
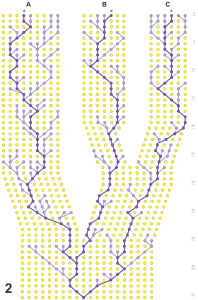
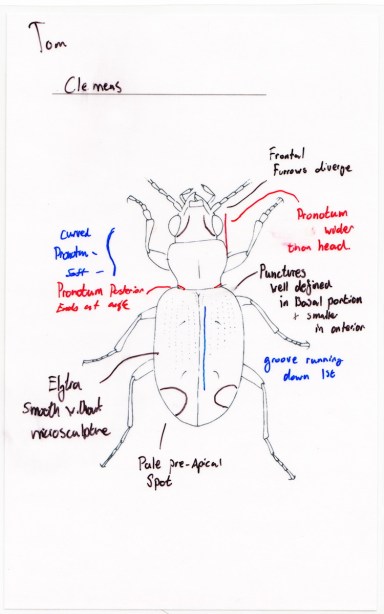
- Thursday, 30 April: In this class students were given blank, laminated cards with an outline of a “generic” Trepanedoris. The students had access to the literature, the knowledge they had gained to date, all the vouchers, and microscopes. Their goal was to make cards showing the diagnostic features of a species, by marking up the card with a sharpie. Each student was responsible for making a card for each of two species. Each species had cards made by two different students, who then had to get together and come up with a single card that synthesized their views about the diagnostic features. It was made more difficult to do as more than half of the species are not described in Lindroth’s (1963) work, so the students had to figure much of it out on their own (with my help, of course). After doing this, students looked at the specimens they sequenced, and made revised predictions about what species they were, and where they would fall in the gene trees.
Week 6
- Tuesday, 5 May: The DNA sequence data is available!!! We download it in class, and process it for the first time as a group. There are four sequencing plates, each containing 96 sequences. Once the data was downloaded, I made initial base calls and contigs for all four plates in about 5 minutes using Mesquite/Chromaseq/Phred/Phrap. I did this on my laptop hooked up to a projector, so that the class could watch as it happened.
I incorporated the 28S sequences from the beetles the students caught in Corvallis and at Klamath Marsh into the existing matrix of 28S sequences, and passed the matrix around to the class.
The class then did a RAxML analysis to get the gene trees, and they each had to see where their four specimens ended up, and whether or not the relationships were as they predicted.
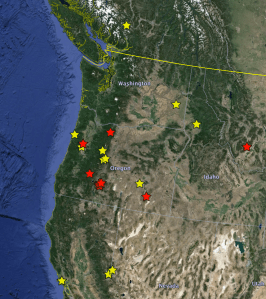
And then I incorporated all of the remaining sequences, including from a large number of specimens from all around western North America, and did analyses for all four genes. This was all done in class on the big screen, and it was very exciting for all of us to see the results live. The most exciting discovery, by far, was that the two specimens we collected at Klamath Marsh that I initially thought (while in the field) were Bembidion “Lost Lake” were not that species at all, but instead another species I had never seen before!
- Thursday, 7 May: I printed out large versions of the gene trees we inferred on Tuesday, and posted them on the walls of the class. After all of the excitement of the previous class, we spent this class sorting Trepanedoris, and mounting other specimens from Klamath Marsh.
Week 7
- Tuesday, 12 May: Lecture and exercises about gene trees and species trees. Mini-lecture on how nomenclature works.
- Thursday, 14 May: Mini-lecture: models and the real world. This was a class with a lot of discussion, and some exercises to help students understand how models are used in science, the nature of hypothesis testing, and estimation. In one exercise, students formed three groups of three, with each group having to come up with coherent answers to the following questions:
- What is meant by a “model of a species tree”
- What is a gene tree?
- A particular model of a species tree has implications as to what we expect the gene trees to look like. Why?
- How can we use this information to infer the species tree?
- How does inferring the species tree allow us to determine what species exist now in nature?
- Friday-Sunday: Weekend field trip to Malheur National Wildlife Refuge
Week 8
- Tuesday, 19 May: DNA extraction from Malheur NWR trip, of specimens chosen by David.
- Thursday 21 May: PCR of Malheur specimens, plus extensive PCR of the gene MSP for previous specimens.
Week 9
- Send plates to GATC sequencing facility
- Tuesday, 26 May: Each student had to do a “Taxonomy Now” presentation, which consisted of their choosing 0ne paper from the literature that described a new species, and telling us about the paper. They had to tell us how many species were described, what evidence was used to claim that the new species were distinct from other species, whether that evidence was convincing, whether the authors had looked at the type specimens of the already described species, etc. Students chose everything from sharks to fungi.
The students also had free time to work on their projects during the class. - Thursday, 28 May: We mounted specimens from the field trips, looked at pictures of type specimens, and did more sorting.
Week 10
- Tuesday, 2 June: We got back the last batch of sequences (about 380 more), and processed them in class. This time the students had to do much of the processing themselves, including inferring the gene trees. They talked in groups about the results, and we talked as a whole. There was much excitement in this class as well, as there were many interesting discoveries made. The most exciting was the confirmation that Shannon’s specimen was not in fact B. canadianum, but was instead an undescribed species that I had never seen before. (I say “confirmation” as after my earlier blog, I did a genitalic dissection, and realized right away that it wasn’t B. canadianum.) We also got for the first time a lot of data from the gene MSP, and that contained a lot of interesting information. We learned a huge amount about Trepanedoris in this class.
- Thursday, 4 June: This was the last class. We were all sad to have it end. At the start of the class, we took out large printed versions of the latest gene trees (which now contain about 220 specimens in them) and taped them up for everyone to look at. The class had to make their final decisions about the species boundaries based upon the gene trees, geographic data, and morphological data. They talked about it as a group – and came up with a very reasonable proposal. As a bonus, we had Nanaimo bars to eat.
Post-class
- Saturday, 6 June: We had the class party this day, including awards to each member of the class. More about these in another post!
- Thursday, 10 June: We got together this day so that we could share projects with each other. The projects were magnificent. More about these in another post!



















Looks like a fun course. The April 30 excersize in particular looked interesting, as students had to synthesize separate takes on the same thing.
They sure didn’t have courses like this back when I was in university!